 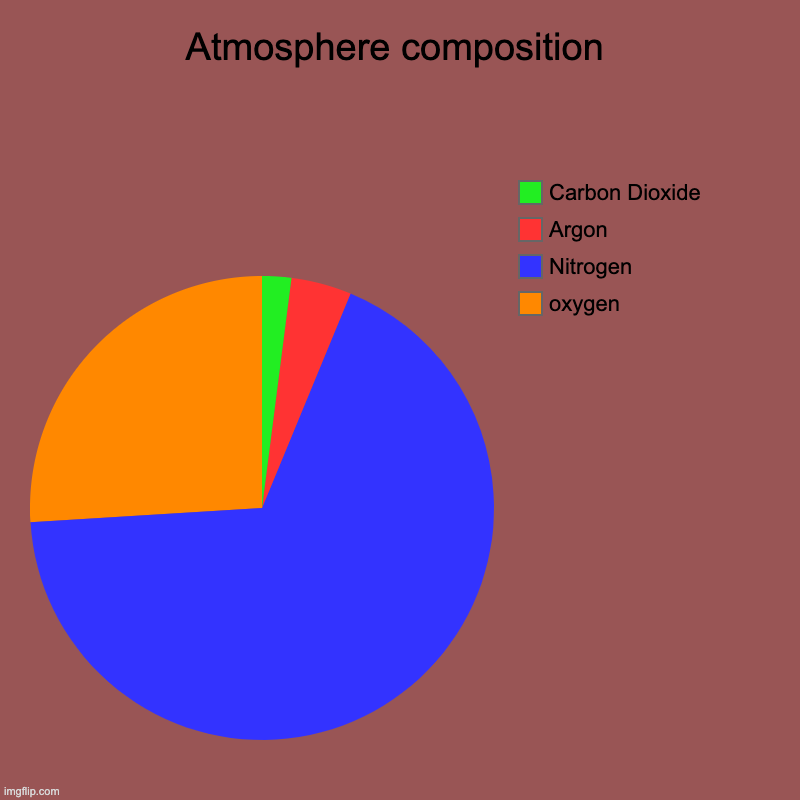
When measuring the heat-trapping greenhouse gases that are causing climate change, such as carbon dioxide and methane, the "laser's" beam is made of infrared light, which has a slightly longer wavelength than the visible light our eyes can see. To study the makeup of the atmosphere, scientists collect some air in a container and then shine what looks like a laser through the sample. These absorption patterns can be used as a kind of chemical fingerprint: by measuring how much radiation a sample absorbs, and at what particular wavelength, scientists can determine which substances are present and in what amounts. This works because all atoms and molecules absorb and emit light at particular wavelengths. A tried-and-true technique in many branches of science, spectroscopy involves shining electromagnetic radiation-which might be visible light, infrared or ultraviolet light, or other types of radiation like x-rays or microwaves-through a sample and studying the effects. The main way scientists measure greenhouse gases in the atmosphere is spectroscopy, says Jesse Kroll, an MIT professor of chemical engineering and civil engineering who specializes in atmospheric composition. So how do researchers get this information? They need this data not only to model and predict climate change, but also to track substances that contribute to pollution and other air quality issues. Scientists regularly measure the gases that make up our atmosphere. 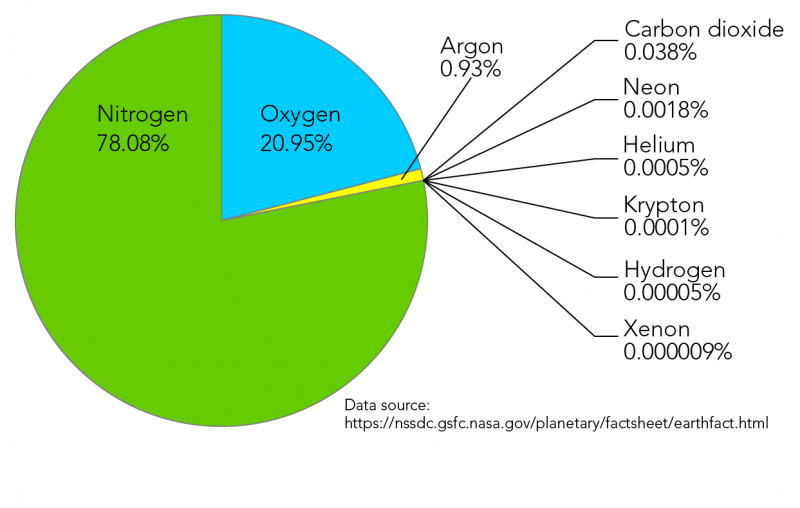
By shining different kinds of light and radiation through air samples, scientists can tell which gases are absorbing or reacting to that light, and in what amounts. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
